Spring 2020 Newsletter
President's Message
 Greetings everyone!
Greetings everyone!
The Sonoran Desert is absolutely gorgeous with abundant wildflowers and the mountains surrounding us, completely green. Hiking trails are sadly not being used to our advantage due to social solidarity in staying at home and social distancing. It’s tough for everyone!
In these unsettling times of a global health crisis, I wish you warm spring greetings. I’ll miss reaching out to many of you personally now that our Annual Meeting has been cancelled. It was a very difficult decision to make the call but during these unprecedented times, it was the only decision to make.
I would first like to acknowledge our Annual Program Committee who worked tirelessly throughout this past year to develop a scientific program that would entice and engage all attendees with all levels of Mohs experience. Nashville 2020 was to be the largest number of registrations on record and for that, we are very proud. Many thanks to our program Chairs, Lindsey Riggs and Norma Anderson and the rest of the committee, Angel Maden, Dan Gong, Dawn Willett, Jeanie Wade, Kathi McAdoo, Robert Tagliaferro, Robin Seaver and Scott Croughan. You’ve all done a tremendous job!
I know that several of you depend on these annual conferences to fulfill your CE requirements. Here’s a list to explore for continuing education. Many are available for free.
- https://www.sakuraus.com
- Webinars : Leica Biosystems
- National Society for Histotechnology - NSH
- CAP Online Education
- Labroots
- Centers for Disease Control and Prevention (CDC) - Laboratory Training
- CDC Public Health Grand Rounds
- Fisher HealthCare → Webinars
My sincere appreciation and thanks to all of speakers and faculty who already had much, if not all of their content developed and ready to present. I’m very grateful for your efforts and hard work that unfortunately will not be presented this year. My hope is that all of you will consider participating by moving your well thought out and constructed content to 2021 as we prepare for the Annual Meeting in Seattle, WA.
We depend on our exhibitors and vendors. There is an extensive amount of time, coordination and dollars spent for these meetings which is all necessary for our conference to achieve its educational goal. We are grateful for their sponsorship and look forward to their continued support.
The Scholarship Task Force took flight this year. It was chaired by Angel Maden and Jeanie Wade. Thank you to both of you for spearheading and finally getting this project off the ground.
Congratulations to Nicole Ryan, our first recipient of the ACMS Annual Meeting Scholarship! The ACMS sponsored scholarship will be awarded annually to an individual who has been a current member of ASMH for at least two years and has never attended the Annual Meeting. The award covers expenses up to $1000 towards meeting registration and extra workshops, hotel, flight and ground transportation. The recipient is recognized at the annual business meeting. At the conclusion of the meeting, this individual is expected to write their experience of the meeting which will be featured in the newsletter. We’ll see you in Seattle, Nicole!
Our Board of Directors experience changes annually by welcoming new faces as well as saying goodbye to others. Our ACMS representatives will transition this year. Dr. Shari Ochoa will be stepping off the Board of Directors as Dr. Daniel Eisen steps on. We extend our great appreciation to both of you for your time and commitment to our organization. You are the voice-of ACMS and a voice- for ASMH. As we have for the last 26 years, we continue to collaborate, so thank you!
Marilyn McCullough will also be leaving the Board and we thank her for her invaluable service and contributions. Dan Gong will be transitioning into his role as President and I look forward to my continued mentorship as I transition to Immediate Past President. Because we have not had an official business meeting and as stated in the Bylaws, the announcement of who you have voted in as your new Vice President and one Director cannot be announced until this happens. Stay tuned for the business meeting date yet to be determined and the platform of how such will happen.
I’d like to recognize Dr. Linda Cesario, Immediate Past President. Her service and commitment to ASMH is exemplary. She will be leaving the Board of Directors after serving 16 years of uninterrupted service. Linda will continue to chair the Mohs Tech training program. She is a well sought after speaker and contributor to the annual program and to the society as a whole so it is our hope that she’ll continue her participation. Thank you Linda for all you’ve done and continue to do for us.
An enormous amount of gratitude is due to our amazing EDI staff. It’s been a huge undertaking for them to undo a done meeting. Thank you to our Executive Director, Mary Randall, and the rest of our dream team, Vanessa Goodman, Mario Ortiz and Dustin Syrjanen. Like many of us, they are experiencing the same navigating challenges as they work remotely from Wisconsin that has a mandatory “Safer at Home” order in place. It’ll take some finesse for them to complete what they need to do for every member and vendor/exhibitor but I know this extraordinary team will make it happen.
During this unusual time, possibly take the opportunity to create a webinar for on-line education or perhaps share uplifting stories of what you and your colleagues are currently experiencing during this global pandemic. Submit your experiences to the newsletter or think about a future session you’d like to present at a future meeting. Use the current situation to focus on how to contribute to uplifting times ahead.
This is my last newsletter as President. We’ve accomplished a lot. Collectively we have achieved some outstanding and unprecedented achievements. ASMH is strong because of the involvement of its membership. YOU are crucial to this organization and what makes it matter.
It has been an honor and a privilege to serve you. I truly enjoy being involved and engaged. I encourage all you to do the same.
Stay well, stay safe. Take good care of yourself and each other.
Mo

M. Maureen Gagnot, HT (ASCP)
ASMH President
Spreading the Word: Advocating for a Profession I Love
Camille McKay, M.Ed. BSHCA, HTL, CLT - Memorial Sloan Kettering Cancer Center
My husband often reminds me that we’ve made it to the proverbial finish line. It is that time in our lives after we’ve raised our children and reached the personal and professional milestones we had set for ourselves when we started our journey together. Now, seeing that “finish line” before me, I find myself with the time, focus, and desire to give back to a profession I love. I have been working in the field of Histology for over thirty years, and particularly in the field of Mohs Technology for the last ten. I have been fortunate throughout my professional career to have a flexible schedule that allowed me to raise my two daughters, now professional adults, and to work my way from technician to technologist, technologist to lead, lead to supervisor, and supervisor to manager. During this journey, I have met many people who served as mentors to me, and I am lucky enough to continue to find mentorship within the organization I work today.
I had a very meaningful mentor early in my own professional development. Dr. Schreiber was that mentor, and through his belief in me, he changed my life. This relationship impressed on me the need to give back and to mentor other people, and I have brought this philosophy to my current position as a Health-care manager for a premier institution.
In the article Overcoming the Silence of Generational Poverty, author D.M. Beegle mentioned that a strong link between mentor support and academic success emerged in her studies of students suffering from generational poverty. Mentors facilitated their understanding of social capital possibilities, expanded their networks of support, and let them know about important resources (Beegle, 2003). Even though I didn’t come from a background of generational poverty, I was raised in a family in which the quest for education was not valued. My mentor helped me to understand that I could overcome this cultural mentality and pursue greater goals.
I recently pursued and completed a Master of Education in Curriculum Instruction, with the goal of utilizing what I have learned through both my profession and my studies to create a well-rounded training program toward the development of Histology professionals.
Not only do I look up to mentors, but I love to mentor people as well. I currently work with colleagues, physicians and administrators to find ways of mentoring the new staff coming in. I recently hired two freshly graduated hires who were trained across both the Mohs and main histology laboratories. The program has been a tremendous success for both the employee and the employer.
The laboratory profession, which includes Lab Medicine, Pathology and Mohs, continues to be under-recognized as a career choice, leading to the current workforce shortage, even as the BLS (Bureau of Labor Statistics) reports that employment of medical laboratory technologists and technicians is expected to grow by 13%, from 330,600 in 2010 to 373,500 in 2020. This translates into about 11,300 laboratory technologist/technician job openings annually through 2018 (including 7,000 through attrition as well as 4,300 new positions). Unfortunately, the programs preparing tomorrow’s laboratory workforce are training only about a third of what is needed to meet this demand. Fewer than 5,000 individuals are graduating each year from accredited training programs. Compounding the problem, the total number of training programs has decreased.
With this information at hand, I continually look for ways to spread the word of a profession that not enough people are aware of. I sit as a board member on both the American Society of Mohs Histotechnology (ASMH) and Goodwin University in Hartford, Connecticut. At Goodwin University, for example, I presented a “train the trainers” program demonstrating how to implement a Mohs program within their own histology curriculum.
I also continue to reach out to local trade schools and have given presentations on both histology and Mohs technology to high school students. I am always searching for ways to share what I have learned within the past thirty years, and to advocate for a profession that I truly love.
References
Beegle, D. M. (2003). Overcoming the silence of generational poverty. Talking Points, 15(1), 11-20.
Retrieved from https://ncte.org/resources/journals/talking-points/
Laboratory Standards. (2018, September 17).
Retrieved from https://wadsworth.org/regulatory/clep/clinical-labs/laboratory-standards
 Preventing Floaters in the Mohs Lab
Preventing Floaters in the Mohs Lab
Melinda Chow, MS, HT(ASCP)CM - Memorial Sloan Kettering Cancer Center
Floaters are microscopic extraneous tissues that sometimes can be found in completed stained slides. The presence of floaters can lead to inaccurate diagnoses, especially when they are cancerous and not matching the rest of the section. It is difficult to completely prevent floaters in our lab. But we can take steps to understand where they come from and how we can prevent them.
The main sources of floaters can be traced from when the specimens first brought to the lab, to microtomy in the cryostat and staining/cover slipping of the slides. Cleaning at each step of processing the specimens plays an important role in the prevention of floaters. It is prudent to clean any extraneous tissues on the specimens with fresh cotton tip applications between each case/patient before relaxing tissues. Some techs wash the specimens in water with bleach to clean/disinfect them. All forceps and blades handling tissues need to be cleaned as tissue fragments can be carried from one specimen to the next.
All blades, the stage where sections are picked up, and anti-rolling plates and brushes used in taking frozen sections in the cryostat should be cleaned and debris free before sections are taken. Tissues have also been showed to be transferred between slides in the H&E stainers. Reagent receptacles in the H&E stainer should be thoroughly cleaned before use.
One should be mindful and use common sense practice in handling slides and coverslips by not touching the slides but to handle the slides by the sides so as not to leave your own cells on the slides. Wearing gloves in handling slides/coverslips are crucial.
Preventing floaters will help avoid misdiagnosis and provide high standard of patient care. As such, cleaning should be incorporated in the standard operating procedures in the lab and every Mohs tech should be trained appropriately to reduce and prevent floaters in the lab.
The ASMH offers customized, hands-on training sessions through its Advanced Mohs Technician Training Program. This on-site training will include lecture and hands-on training on topics such as mapping and inking, embedding, staining, cryosectioning, CLIA regulations, troubleshooting, and more. This program is designed to enhance both the performance and quality of work of established Mohs technicians - those with at least six months of experience in Mohs. Please note that this training is not an official Histotech Certification program, nor is it intended for techs with no experience.
Thursday, June 18 - Friday, June 19, 2020
8:30 am - 3:00 pm
Avantik Academy Mohs Training Campus, 19 Chapin Road, Building C, Pine Brook, NJ, 07058
Thursday, September 17 - Friday, September 18, 2020
8:30 am - 3:00 pm
Avantik Academy Mohs Training Campus, 19 Chapin Road, Building C, Pine Brook, NJ, 07058
- ASMH members: $750 / Non-members: $1,000
- Non-member registration will include ASMH membership through 2020 for techs working for ACMS member surgeons
- $50 discount per trainee for practices sending more than one tech
Upon completion, trainees can claim 12 CEUs through the National Society for Histotechnology and will receive a certificate of completion from the ASMH/ACMS. For more information about the Advanced Mohs Technician Training Program or customized training opportunities, click here or contact the ASMH office at (414) 918-9813 or info@mohstech.org.
 Troubleshooting H&E Staining: Brown Staining
Troubleshooting H&E Staining: Brown Staining
Norma L. Anderson, HT(ASCP) - University of Washington Cancer Center
In 2014 I submitted an article about Eosin Bleeding. I discovered that xylene substitutes absorb atmospheric moisture in humid climate. I thought that I had finally solved my staining problem by using xylene after 100% alcohol followed by xylene substitutes. I was able to coverslip from the xylene substitute; I had no more eosin bleeding.
Issues with a very common Hematoxylin and Eosin stain continue to arise. In November of 2019 I started having a problem with Brown Staining. The Brown Staining was seen throughout the section; it was in the dermis and in some of the basal cells. A Brown Staining is common when tissue is not well dehydrated. Even though my slides were getting adequate dehydration my slides had this staining problem. This Brown Stain was very sparse; the staining was maybe on one or two cases per day and not on every section.
To troubleshoot the problem, I checked my staining solutions to make sure that everything was up to date. Even though my solutions were all good I ordered new hematoxylin to try. Another thought I had was that my stainer was the problem. I use a Leica ST4020; the slides on this stainer agitate in an up and down motion. I wondered if this motion was not sufficient for alcohol solutions. I tried adjusting the staining time and number of dips on the stainer. I also ordered new slide clips for my stainer to see if it would help. None of these changes made a difference; there was still Brown Staining.
In March of 2020 I remembered that I was currently not using xylene in my stain line. I had stopped using the xylene since our lab humidity was back to normal range. There was no obvious sign that the eosin was bleeding out of the sections so I didn’t make the association that omitting xylene was the problem.
So once again I added the xylene back to my stainer after three 100% alcohols; xylene substitute following the xylene. Once again, I still cover slipping from xylene substitute. Inadequate dehydration not only causes eosin bleeding but Brown Staining as well. Adding xylene to my stain line has solved both problems in my lab.
Reflectance Confocal Microscopy and the Impact on Mohs Histotechnology
Gil Genna Manuel, HT(ASCP) & Brandon Possum, HT(ASCP) - Memorial Sloan Kettering Cancer Center; Acknowledgement to Dr. Saud Aleissa for sharing his expertise
What is Reflectance Confocal Microscopy (RCM)?
Reflectance Confocal microscopy (RCM) is an exciting and innovative new tool that is increasingly being employed by dermatologists across the world. It is FDA approved for the diagnosis of different skin lesions, including non-melanoma skin cancers. While a skin biopsy with histopathology is the gold standard for diagnosis, it is invasive and occasionally painful.[1] RCM is a non-invasive, quasi-histological optical device capable of obtaining real-time, horizontal section images of the skin up to 200-250 μm in depth. In addition to helping determine if a questionable skin lesion is benign or malignant, it may also help determine the extent of the lesion, beyond what the naked eye can see. This can help with pre-surgical assessment and planning, margin mapping and monitoring response to non-surgical treatment in a completely non-invasive manner.
What Does this mean for Mohs Histotechnology?
Since RCM technology is a growing field of interest, studies can be carried out with collaboration between the Mohs surgeon and the Mohs technician. For instance, at Memorial Sloan Kettering Cancer Center, a correlative study was performed to assess the presence of residual basal cell carcinoma after a conventional biopsy. Lesions were imaged by RCM prior to Mohs surgery and additional, deeper sections were obtained and corelated to the corresponding RCM image. This was achieved by melting down the mounting medium of the Mohs layer and re-embedding the tissue on a vertical plane instead of enfacing. Having the tissue oriented on a vertical plane had results similarly to how a skin biopsy appears. Next, multiple slides were created by taking sections at intervals of 80 microns in between each section until the tissue was exhausted. When reviewed, if tumor was present on any of the sections on the additional slides, it proved a successful correlation to what was found in the imaging.[2] This study was a good example of the kind of collaboration that Mohs surgeons and histotechnologist can achieve. RCM is still an area of dermatology that continues to evolve, and its application continues to increase. While there is associated cost, training, and additional patient time required, these can be offset by increasing efficiency and planning ultimately enhancing the patient experience. While Mohs surgery remains the gold standard for treatment of non-melanoma skin cancer and Mohs histotechnicians are indispensable, RCM continues to be helpful addition to the Mohs armamentarium.
References
1. Malhovy, Josep et al. Acta Derm Venereol (2017). Dermoscopy, Confocal Microscopy and Other Non-Invasive Tools for the Diagnosis of Non-Melanoma Skin Cancers and other Skin Conditions. NCBI. https://www.ncbi.nlm.nih.gov/pubmed/28676883.
2. Navarrete-Dechent C, Cordova M, Aleissa S et al. Reflectance confocal microscopy confirms residual basal cell carcinoma on clinically negative biopsy sites before Mohs micrographic surgery: A prospective study. J Am Acad Dermatol 2019;81:417-26.
Education/Professional Development
Melinda Chow, MS, HT(ASCP)CM - Memorial Sloan Kettering Cancer Center
As the saying goes, “If you fail to plan, you plan to fail”. I am glad I planned and did what I wanted to do last summer. I embarked on a 2-week cultural/foodie and sightseeing trip to Spain and Portugal. I learned new culture, new language, new food and, most of all, the sightseeing was spectacular. It was one of the most enjoyable trips I have ever taken.
The following are some of the things you can plan to do on the work front for your educational and professional development:
- Get your HT or HTL certification, as more employers are making it a requirement for employment. You can get the necessary information on the ASCP Website.
- Obtain the state license, if required, in the state you plan to work.
- Pursue a college degree to advance your career.
- Learn new skills/techniques. This is how I first learned my cryotomy skills. The company I was working for wanted to implement a new test that required cutting frozen sections. I volunteered to learn and took on responsibilities for this new test. I acquired this new skill which helped me in getting my Mohs tech position.
- Enroll in one of the Mohs Technician Training Programs to improve your current skills.
- Contribute knowledge by writing articles for the ASMH Newsletter. I know writing the first one is hard but once you get started, it will get easier. Put on your thinking cap and I am sure you will find something to write. Everyone has a story to tell.
- Participate in workshops and/or prepare for a talk at the ASMH Annual Meeting.
- Fulfill the required annual 12 CEUs by attending the ASMH Annual Meeting or by taking complimentary webinars offered by Leica Biosystems or Sakura Finetek.
- Volunteer to help in the clinic. I accompany my Mohs surgeon when he examines patients, especially female patients for skin checks. Your Mohs surgeon will appreciate your help and recognize your enthusiasm in the workplace.
These are some of the things you can do to advance your education and career and develop yourself professionally. Don’t feel stuck in a dead-end job as there are limitless opportunities for you to explore and advance.
Take Advantage of the Mohs Surgery Career Center

Visit the online Mohs Surgery Career Center to find or post open histotech positions. ASMH members can search openings, post resumes and connect with employers. Contact info@mohstech.org for help accessing member pricing.
Indeterminate Surfaces: Processing Non-Keratinized Epithelium in the Mohs Lab
William H. Phillips - Memorial Sloan Kettering Cancer Center
Nail Bed
As a Mohs technician, I have found that the most challenging cases to process involve tissue specimens that lack a keratinized epithelial layer. Epithelium is a thin layer of tissue that lines the outside of organs. For our purposes as Mohs techs, the epithelial layer we encounter the most during tissue processing is the epidermis, which covers the skin. Not all epithelium is the same, it has different characteristics depending on the needs of the organ it covers. The skin needs to be guarded against abrasion, so cells in the epidermis, as they mature and move towards the surface, undergo a process called keratinization. When they reach the uppermost layer of the epidermis, the cells form a hardened protective layer of keratin called the stratum corneum. Not all skin surfaces need a barrier to protect against abrasion, so not all skin epithelium is keratinized. One example is the inside of the eyelid. The inner eyelid (the posterior lamella) delicately cradles the eyeball and must be lubricated by substances secreted through a mucosal epithelial surface, called the conjunctiva, which is not keratinized.[1] For any Mohs tech who has encountered the complexities of processing conjunctival eyelid, the lack of a keratinized layer makes clear why this issue is so important for the Mohs tech. The stratum corneum is a visible marker that the Mohs tech can use to identify epidermis, during grossing, embedding (as it freezes and turns almost white) and sectioning. Without the stratum corneum it becomes very difficult, at every juncture of the process, to assess whether or not you have captured the epidermis for microscopic inspection by the surgeon.
In the case of nail bed, there is a keratinized layer, the nail plate, but it is typically removed by the surgeon before processing. So, in this context, with the nail plate removed, the epithelium of the nail bed approximates a non-keratinizing epithelium and offers the same challenges.
Depending on what kind of tissue you’re processing, the epithelium may present in different ways. For instance, given their unique anatomical characteristics, conjunctiva and urethra need different grossing and embedding techniques to obtain full epithelium, than you may use with a specimen taken from the cheek. It is helpful in these cases to have a basic grasp of the anatomy in order to understand what you need to do as a Mohs technician to obtain full representation of the base and lateral margins. The anatomy dictates which technique will be used. For a nail case, the epithelium is the surface of the nail bed, underneath the nail plate.
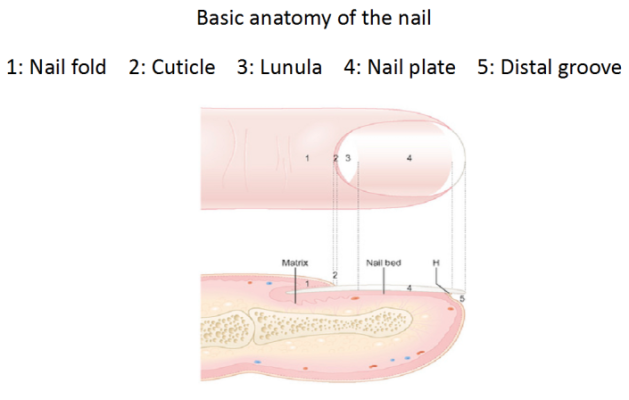
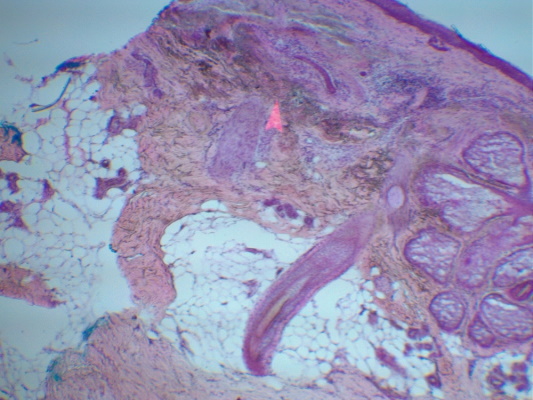
The primary structures are the nail fold, cuticle, lunula, nail plate, distal groove, and the nail bed, underneath the plate.[2] The nail plate grows from the matrix, which is located at the proximal end of the nail. The lunula, that little white crescent, is the distal part of the matrix. You can see a few of these features on a slide we did for a recent case.
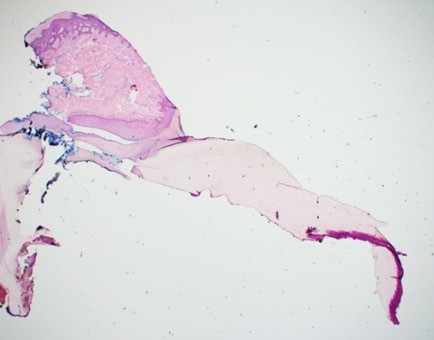
On this vertical section you can see the nail fold above, the nail plate below, and the cuticle sealing the area between the fold and the plate. Note also that on the lower right is nail bed epithelium attached to nail plate. As you can see, one nail section can pose a host of processing problems. Nail fold can separate from the rest of the tissue during grossing, nail plate can fall off the slide like cartilage, and nail bed epithelium is extremely tricky to fully represent.
As with any tissue specimen the Mohs tech needs to identify the lateral periphery, the surface, and the base. The nail bed epithelium lies where the surface meets the lateral periphery. In terms of processing difficulty, it is the surface of nail bed epithelium that poses the greatest challenge. It may only be a few cells thick, and when the nail is removed some of those cells can remain attached to the nail. Nail bed tissue is brittle, so you can’t fold your peripheral margin down, and your specimen will be thin, so relaxing will probably not be an option, and the tissue texture doesn’t favor it anyway. I think due to the fragility of the tissue, you don’t get a great bevel either. So, nail bed is a prime candidate for the double-embedding technique.[3] This is when you embed the base first without trying to obtain the lateral margin; you cut a few sections of the base on one slide, then melt the block down and re-embed the piece on its vertical edge. Now, on a second slide, take vertical sections of the lateral margin until you get full epithelium. You may want to re-ink between the stages and your surgeon may request separate maps, one for the base and one for the lateral margin.
One additional technique that I have found helpful when grossing nail bed, is to color the surface of the bed with a surgical marker. This ‘mock’ stratum corneum will help you to identify the surface of the lateral margin during embedding, and when sectioning it will give you a clear indication that you are fully representing the nail bed epithelium.
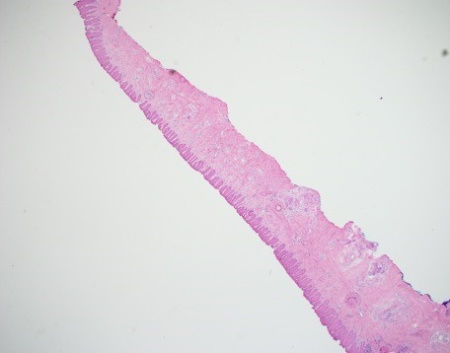
Here is a vertical section of nail bed epithelium from a recent nail case. By using the double-embedding technique to capture the surface of the lateral margins, and by coloring the nail bed epithelium with a surgical marker, you can give yourself a very good chance of succeeding in what can otherwise be daunting circumstances.
References
1. http://library.open.oregonstate.edu/aandp/chapter/4-2-epithelial-tissue/
2. https://basicmedicalkey.com/diseases-of-the-nails/
3. Karen, Julie K., Hazan, Carole E., Tudisco, Marie, Strippoli, Barbara, Nehal, Kishwer S. A Modified Technique for Processing Mohs Wedge Excisions. (Report). Dermatologic Surgery. 2009;35(4).
Webinar Topics Needed
ASMH recognizes that many members rely on CEU credits received at the Annual Meeting to fulfill their annual credit requirements. We are exploring other options for you to continue to learn and receive the credit hours you need, and we plan to provide you with additional opportunities in the near future.
We will be developing a series of webinars related to Mohs histotechnology. What topics do you wish to see covered in a webinar? Let us know! Are you interested in sharing your skills as a webinar presenter? Let us know this, as well!
Please complete this brief survey and share your thoughts. Thank you!
Submit an Article for the ASMH Summer Newsletter
We're seeking your ideas and submissions for the ASMH Summer e-Newsletter! Please consider your recent Mohs lab experiences; are there things you’ve done or learned that your fellow members may want to know about? Think of successes, tips, tricks or anything else that helps advance the work of Mohs technicians.
Here are a few general ideas to get you thinking:
- Achievements: Any recent accomplishments or recognition?
- Community Engagement: How do you spread the word about ASMH & Histotechnology?
- Education and certification: How did you or will you prepare for HT or HTL certification?
- Trends: How do you and your colleagues keep up with what’s new in the field?
- Jargon: What technical shorthand have you picked up, used or created?
- Equipment: Any new tools, technology or tips for using them in your lab?
- Quality Control: How do you deal with standards, enforcement, charts and more?
- Records: Paper or electronic, what do you do to keep clear, organized records?
- Safety: How do you ensure safety around chemical and biological hazards in your lab?
The target word count for most pieces is between 300-600 words. As a reference, 600 words is less than a page of single-spaced text! If you feel a particular subject bears more in-depth examination, that's okay too. Also, think of what visuals you could provide--photos, graphs, charts, screenshots, infographics, etc.
Questions? Have an idea of something you'd like to submit? Email dsyrjanen@mohstech.org with your ideas or submissions! As always, thank you for your involvement in the ASMH!
Member Resources
ASMH Workforce Survey
One of the benefits of ASMH membership is the ability to contribute to ASMH's biennial Workforce Survey. Data obtained from this survey will not only help examine industry standards and trends, but will assist you and other Mohs technicians in determining if you are paid competitively. When you know what the industry standard is, you can better negotiate your salary with your current employer during the performance appraisal process. This information can also assist you with future job search efforts.
To view the results of the 2019 Workforce Survey, log in to the Members page on the ASMH website.
ASMH Bibliography - Newly Added COVID-19 Resources
The ASMH bibliography is your resource and we strongly encourage you to explore it. Efforts will be made to keep the articles up to date and relevant to current best practices while keeping the scope wide enough to interest the broadest of minds. It is our intention to cover subjects ranging from basic lab techniques, Mohs embedding, cryotomy, routine and immunohistochemical staining, lab safety, ethics and work flow. This is a continually evolving project that will be guided by your feedback, suggestions and more importantly, your contributions, so please share interesting articles and send topic suggestions to info@mohstech.org.
Go to the bibliography and find new COVID-19 resources: Laboratory Biosafety & Coronavirus Disinfection in Histopathology.
Member Marker

The American Society for Mohs Histotechnology has a rich tradition of dedicated members that work hard to keep the ASMH true to the original commission; to uphold the importance of training, collaboration and consistency, while working in conjunction with the American College of Mohs Surgery to ensure that the high standards set by Dr. Mohs continue to be met.
Members are encouraged to download, and use this graphic in your email signature, on your social media profile or added to your professional online presence. Please remember that you may not, in any way, alter, manipulate or change this graphic. Contact info@mohstech.org should you have any questions about proper use.
Note to ASMH Newsletter Readers:
Reference to any specific commercial product, process, or service by trade name, trademark, manufacturer, or otherwise throughout this e-newsletter, does not constitute or imply its endorsement, recommendation, or favoring by the American Society for Mohs Histotechnology. The views and opinions of authors expressed do not state or reflect those of the American Society for Mohs Histotechnology.